 |
|
|
|
 |
|
|
 |

|
 |
 |
 |
 |
 |
 |
 |
Creating Point Defects |
Our lab has developed a suite of techniques for creating and studying defects in SWNT conductors.
To create defects, we primarily use an electrochemical technique that gives us single-defect control.
Our work begins with individual, electrically connected SWNTs in a backgated, field-effect transistor
(FET) geometry. Using this type of device as an electrochemical working electrode, we can apply a voltage
between a SWNT and any surrounding electrolyte. At low bias, most electrolytes noncovalently dope a SWNT
by accumulating ions near its surface. Using higher bias, we can drive covalent oxidation or reduction
reactions between the SWNT and its environment.
SWNTs are usually excellent electrical conductors, and when an oxidation event occurs it produces a
noticeable decrease in the two-terminal conductance. In fact, multiple discrete drops in conductance
indicate the step-by-step oxidation of a SWNT in real time as it gradually degrades into a completely open
circuit. For our work investigating single defects, we simply bias the working electrode close to the
threshold for a particular chemical reaction, and then turn off that bias (using a trigger) when the desired
conductance is reached.
A useful aspect of this technique is that the same SWNT can be studied before and after a
particular chemical modification, without any other changes to its intrinsic scattering or
contact resistance. By studying the same device with and without a defect, for example,
we gain precise knowledge about that defect’s electrical consequences.
By changing the electrolyte, we can electrochemically promote a variety of chemically distinct
defects, each of which can have different consequences.
|
 |
Publications |
Atomistic Oxidation of a Carbon Nanotube in Nitric Acid
Y. Kanai, V.R. Khalap, P.G. Collins & J.C. Grossman,
Phys. Rev. Lett. 104, 066401 (2010).
Scaffolding Carbon Nanotubes into Single-Molecule Circuitry
B.R. Goldsmith, J.G. Coroneus, J. Lamboy, G.A. Weiss & P.G. Collins,
J. Mat. Res. 23, 1197-201 (2008).
Mechanism-Guided Improvements to the Single Molecule Oxidation of Carbon Nanotube Sidewalls
J.G. Coroneus, B.R. Goldsmith, J.A. Lamboy, A.A. Kane, P.G. Collins & G.A. Weiss,
ChemPhysChem 9, 1053 (2008).
Conductance-Controlled Point Functionalization of Single-Walled Carbon Nanotubes
B.R. Goldsmith, J.G. Coroneus, V.R. Khalap, A.A. Kane, G.A. Weiss & P.G. Collins,
Science 315, 77 (2007).
Chemically Induced Conductance Switching in Carbon Nanotube Circuits
J. Mannik, B.R. Goldsmith, A. Kane & P.G. Collins,
Phys. Rev. Lett. 97, 16601 (2006).
|
 |
Acknowledgements |
| This research is financially supported by the NSF. |
 |
|
|
|
 |
|
|
|

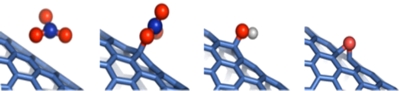
Schematic of doping, oxidation, and reduction at a single site.

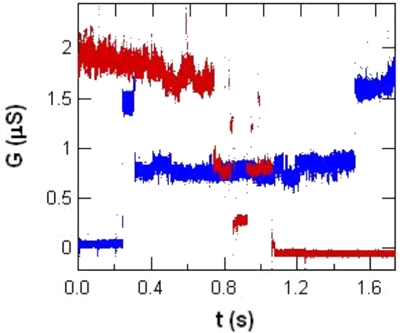
Overlaid time traces of electrochemical oxidation (red) and subsequent reduction (blue).

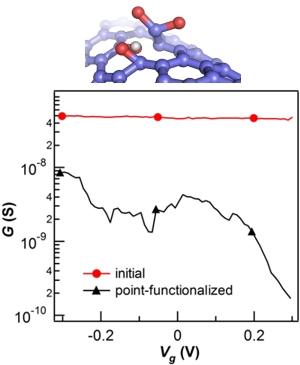
Cartoon of a -COOH defect and its electrical effect on a metallic SWNT.
|
 |





